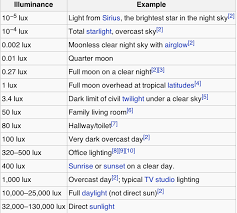
-
Posts
980 -
Joined
-
Last visited
-
Days Won
15
Content Type
Profiles
Forums
Calendar
Downloads
Gallery
Everything posted by JJ the Gardener
-
From the album: Hempyfan
-
From the album: Hempyfan
-
From the album: Hempyfan
-
From the album: Hempyfan
-
From the album: Hempyfan
-
From the album: Hempyfan
-
From the album: Hempyfan
-
From the album: Hempyfan
-
From the album: Hempyfan
-
From the album: Hempyfan
-
From the album: Hempyfan
-

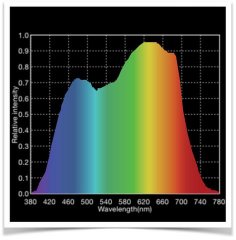
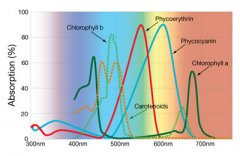
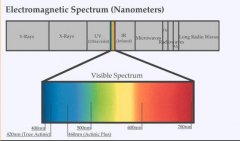
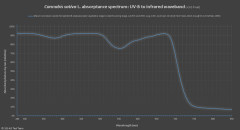
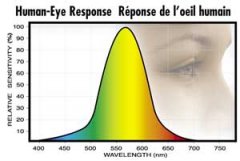
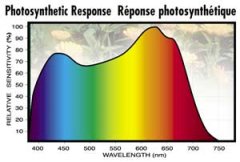
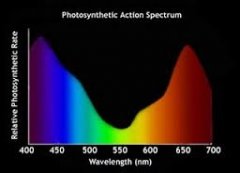
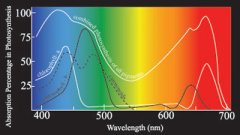
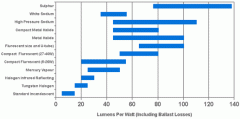
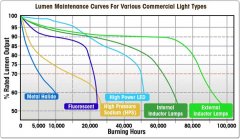
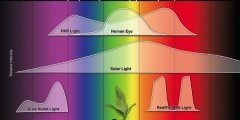
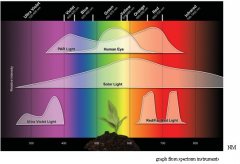
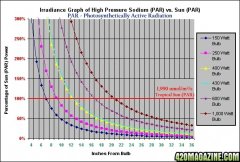
lighting-par-sun-eye-comparison-chart-600x300.jpg
JJ the Gardener posted a gallery image in Member Albums
From the album: Hempyfan
-
From the album: Hempyfan
-
As California legalizes pot, laws collide at US checkpoints Elliot Spagat, Associated Press,Associated Press https://www.yahoo.com/news/california-legalizes-pot-laws-collide-us-checkpoints-163506609.html PINE VALLEY, Calif. (AP) -- California legalizes marijuana for recreational use Monday, but that won't stop federal agents from seizing the drug — even in tiny amounts — on busy freeways and backcountry highways. Marijuana possession still will be prohibited at eight Border Patrol checkpoints in California, a reminder that state and federal laws collide when it comes to pot. The U.S. government classifies marijuana as a controlled substance, like heroin and LSD. "Prior to Jan. 1, it's going to be the same after Jan. 1, because nothing changed on our end," said Ryan Yamasaki, an assistant chief of the Border Patrol's San Diego sector. "If you're a federal law enforcement agency, you uphold federal laws." The checkpoints, located up to 100 miles (161 kilometers) from Mexico, are considered a final line of defense against immigrants who elude agents at the border. They also have been a trap for U.S. citizens carrying drugs, even tiny bags of marijuana. About 40 percent of pot seizures at Border Patrol checkpoints from fiscal years 2013 to 2016 were an ounce (28 grams) or less from U.S. citizens, according to a Government Accountability Office report last month. California's new law allows anyone 21 and over to carry up to an ounce. The Border Patrol operates 34 permanent checkpoints along the Mexican border and an additional 103 "tactical" stops, typically cones and signs that appear for brief periods. Ronald Vitiello, acting deputy commissioner of parent agency Customs and Border Protection, called drug seizures an "ancillary effect" of enforcing immigration laws. Motorists typically are released after being photographed and fingerprinted. They generally aren't charged with a crime because prosecutors consider them low priority. The clash between state and federal marijuana laws played out on a smaller scale near the Canadian border in Washington after that state legalized marijuana in 2014. California is a far busier route for illegal crossings with many more agents. State and federal marijuana laws have conflicted since California became the first to legalize marijuana for medical use in 1996. Next week, California will be among seven states and Washington, D.C., with legal recreational pot. U.S. Attorney General Jeff Sessions, a staunch opponent of legalization, said last month that he was taking a close look at federal enforcement, suggesting a tougher stance than President Barack Obama's administration. At highway checkpoints, Border Patrol agents look for signs of nervous drivers, like clutching steering wheels and avoiding eye contact and interrupting when passengers are asked to state citizenship. Some panicked drivers make a U-turn when they spot the checkpoint, a dead giveaway. One recent morning on westbound Interstate 8 about 40 miles (64 kilometers) east of San Diego, an agent standing outside a booth under a large white canopy stopped drivers for a few seconds to ask their citizenship or waved them through after peering inside. In about an hour, three raised enough suspicion to be ordered aside for a thorough vehicle search. A dog discovered a marijuana stash about the size of a thumbprint inside the pickup truck of a man with Arizona license plates who was taking his elderly uncle to a hospital appointment. It would have taken up to an hour to process the arrest, so agents released him after seizing the pot and warning it was illegal. "I didn't know that, sorry," the driver said, walking to his truck after waiting on a bench a few minutes while the dog searched. The animal sniffed something in another car but found nothing in the seats or trunk. The apologetic driver said she smoked marijuana a week earlier, implying the odor lingered. The Pine Valley checkpoint, amid oak- and chaparral-covered mountains on the main route from Arizona to San Diego, gets busy with drivers returning from weekend getaways but is less traveled than others. Agents say a checkpoint on Interstate 5 between San Diego and Los Angeles can cause a 4-mile (6.4-kilometer) backup in 90 seconds during peak hours. The government faces pushback over checkpoints. Some residents complain about delays and trespassers trying to circumvent checkpoints — some even dying from heat and exhaustion. Motorists who consider them a privacy invasion steadfastly refuse to answer questions and post their test encounters on YouTube. Border Patrol officials insist they are effective. Without them, Vitiello said, smugglers would have open passage to cities like Phoenix and Albuquerque, New Mexico, once past the border. The U.S. Supreme Court ruled in 1976 that agents can question people at checkpoints even without reason to believe anyone in the vehicle is in the country illegally and don't need a search warrant. Michael Chernis, an attorney who represents people charged with marijuana crimes, believes checkpoint seizures are a waste of resources but acknowledged the government is empowered. "The bottom line is, there's absolutely no protection against federal interaction when it comes to adult use," he said.
-
Sessions rescinds letter warning local courts about fees and fines imposed on poor people http://www.abajournal.com/news/article/sessions_rescinds_letter_warning_local_courts_about_fees_and_fines_imposed BY DEBRA CASSENS WEISS POSTED DECEMBER 22, 2017, 9:26 AM CST Updated at ABA at 1:05 p.m. with statement from ABA President Hilarie Bass. Updated: Attorney General Jeff Sessions has rescinded a “Dear Colleague” letter that warned state and local courts about constitutional concerns regarding fees and fines imposed on poor defendants. Sessions rescinded the letter along with 24 other documents he found “unnecessary, inconsistent with existing law, or otherwise improper,” according to a statement. The Washington Post and BuzzFeed News have stories. The letter, issued in March 2016 by the U.S. Justice Department, had urged courts to review procedures regarding fines to make sure they comply with “due process, equal protection and sound public policy.” The letter said courts shouldn’t incarcerate a person for nonpayment without first determining whether the person is indigent and whether the failure to pay is willful. Sessions’ statement said he has “ended the longstanding abuse of issuing rules by simply publishing a letter or posting a web page.” Congress has provided for a regulatory process “and we are going to follow it,” Sessions said. “Therefore, any guidance that is outdated, used to circumvent the regulatory process, or that improperly goes beyond what is provided for in statutes or regulation should not be given effect,” he said. ABA President Hilarie Bass released a statement on Friday expressing disappointment in the decision to rescind the fees and fines, and asks the Justice Department to reconsider. “These monetary punishments do nothing to protect the community while placing an unfair and unjust burden on people of lesser means,” the statement said. “Fees and fines that do not take into account a defendant’s ability to pay lead to the criminalization of poverty.” Fees assessed for minor infractions, such as traffic tickets, can “spiral into thousands of dollars” and lead to unnecessary jailing of those unable to pay, the statement also said. “Bail set without consideration of financial circumstances can lead to detention of the poorest rather than those who are the most dangerous or those posing the highest flight risks as intended. … If we, as a country, are to live up to the ideal of equality under the law, then there cannot be a price on justice,” the statement noted. Updated at 1:05 p.m. with statement from ABA President Hilarie Bass.
-
Top Florida court to decide whether ticket-fighting startup engages in unauthorized law practice BY and from ABA DEBRA CASSENS WEISS POSTED DECEMBER 20, 2017, 7:45 AM CST http://www.abajournal.com/news/article/top_florida_court_to_decide_whether_ticket_fighting_startup_engages_in_unau The Florida Supreme Court is being asked to decide whether a ticket-fighting startup is engaged in unauthorized law practice and is misleading consumers. The Florida Bar voted earlier this month ask the court to decide the issues and issue an injunction against the startup, TIKD Services, report the Daily Business Review (sub. req.) and the Miami Herald. TIKD Services developed an app called TIKD in which drivers who receive tickets upload their tickets, pay TIKD a fixed price, receive a lawyer’s services through the app to defend the tickets, and get a guarantee from TIKD that the tickets will not cost any more money, no matter the outcome. TIKD Services filed a federal lawsuit in November that claims the Florida Bar and a competitor launched a coordinated attack to drive it out of business. TIKD is represented in the suit by Ramon Abadin, a former Florida Bar president. The bar has filed a motion arguing that Abadin should be disqualified from representing TIKD Services because he received privileged communications as a bar official that is relevant to the suit.
-
Another lawsuit makes racketeering claims against state-legal marijuana business Another lawsuit has been filed alleging Racketeer Influenced and Corrupt Organizations Act claims against a marijuana business, Bloomberg Businessweek reports. http://www.abajournal.com/news/article/another_lawsuit_makes_racketeering_claims_against_state_legal_marijuana_bus BY LORELEI LAIRD article from ABA Journal POSTED DECEMBER 15, 2017, 8:00 AM CST In Crimson Galeria Limited Partnership v. Healthy Pharms (PDF), the owners of four buildings in Cambridge, Massachusetts alleges that a nearby marijuana business, Healthy Pharms, is a “conspiracy to sell marijuana” that damages the value of the plaintiffs’ property. The plaintiffs also argue that related businesses, the marijuana business’s bank and various government agencies are part of the conspiracy under RICO because they enable and encourage marijuana businesses in violation of federal law. “Extensive evidence shows that marijuana retail sale is an odorous and stigmatized activity and that foul smelling, stigmatized activities reduce nearby property values,” the complaint says. “In light of this evidence, it is clear that defendants’ operation of a marijuana dispensary … diminishes the market value of plaintiffs’ abutting and nearby properties.” It’s at least the fourth RICO lawsuit from a neighbor of a marijuana business, and the third from the same law firm. As the ABA Journal reported in November, one of those cases went to the Denver-based 10th U.S. Circuit Court of Appeals, which ruled in June that those plaintiffs could go forward with property damage claims based on the strong smell of a marijuana farm. Strong smells are part of the complaints in Crimson Galeria, which also alleges that having Healthy Pharms for a neighbor could attract theft because of the large quantities of drugs. Because the plaintiffs are commercial property owners in Cambridge’s busy Harvard Square, they also argue that having a marijuana business for a neighbor creates a stigma that will reduce property values. To document this, plaintiffs hired an appraiser. “People buy property and rent in plaintiffs’ buildings because they want to operate their businesses in a pleasant and historic area, and the Plaintiffs’ land is less suitable for those uses due to the … dispensary,” it says. Like the other cases, this one hinges on the fact that most violations of the Controlled Substances Act automatically violate RICO. This permits the plaintiffs to include as defendant co-conspirators a variety of businesses and people associated with Healthy Pharms, including its landlords and its bank. The complaint also includes several government agencies, saying they are part of the conspiracy because they regulate, tax and tolerate Healthy Pharms. “The people of Massachusetts are free to advocate for a repeal of this federal criminal prohibition, but they must do so through their elected representatives in Congress,” the complaint says. The plaintiff landlords are represented by Cooper & Kirk, a Washington, D.C. law firm a law firm aligned with conservative causes that also brought two of the three other lawsuits. One of those settled; the other, which went to the 10th Circuit, is scheduled for trial in July of 2018. The third, unrelated case has motions to dismiss pending in Oregon federal court. Healthy Pharms co-owner Nathaniel Averill told Bloomberg that the lawsuit has already made it harder to find investors. University of Denver law professor Sam Kamin told the Journal in November that this could be the real purpose of the lawsuit. “The [lawsuits] may or may not succeed, but they have the opportunity to inflict great costs along the way,” he said.
-
- racketeering
- lawsuit
-
(and 3 more)
Tagged with:
-
China's CCTV surveillance network took just 7 minutes to capture BBC reporter Jon Russell,TechCrunch It took Chinese authorities just seven minutes to locate and apprehend BBC reporter John Sudworth using its powerful network of CCTV camera and facial recognition technology. This wasn't a case of a member of the media being forcibly removed from the country. The chase was a stunt set up to illustrate just how powerful and effective the Chinese government's surveillance system can be. It's a stark example of the type of monitoring that China has invested heavily in over recent years with the aim of helping police do their job more efficiently. Such systems are also used in private organizations, for example to monitor workers and processes in factories, but government critics have warned of the potential for abuse in the hands of the state. China has the largest monitoring system in the world. There are some 170 million CCTV cameras across the country, and that's tipped to grow more than three-fold with 400 million more set to be installed by 2020. Beyond the sheer numbers of lookout points, China is harvesting information with a new-found focus on intelligence. The government also works with facial recognition and AI companies, such as unicorn Face++, which can pour through data to extract meaningful information such as faces, ages, registration plates and more. The full video of Sudworth's 'capture' is on the BBC website, with a snippet is below -- hat-tip The Next Web. pic.twitter.com/vLGQYN7ZB9 In China's surveillance state, a reporter's game of hide and seek had a sinister edge http://www.newsweek.com/tasked-trying-remain-undetected-long-possible-sudworth-filmed-himself-selfie-747843 BY CHRISTINA ZHAO ON 12/14/17 AT 6:54 AM China currently boasts the largest monitoring system in the world, with approximately 176 million CCTV cameras in public and private hands. According to The Wall Street Journal, China will add another 450 million cameras by 2020. The U.S., by comparison, has around 50 million. CCTV is also used in China by private organizations to monitor workers and mine human data, a practice that has attracted criticism from activists as an abuse of human rights. China has no enforceable protections for privacy rights against state surveillance, reported Human Rights Watch (HRW). “Until China has meaningful privacy rights and an accountable police force, the government should immediately cease these efforts,” said Sophie Richardson, China director at HRW. Officials told Sudworth that only criminals need to fear the technology, but recent reports suggest that the software has been used to monitor and intimidate ethnic minorities like the Uighurs in western China. Beyond the cameras, China has also been gathering information by using new facial recognition intelligence and working with artificial intelligence companies, which can quickly shift through data to extract information on people’s faces, ages, registration plates and more. Facial recognition cameras and software are also being used in China for routine activities, such as gaining entrance to a workplace, withdrawing cash from an ATM and unlocking a smartphone, reported WSJ. A KFC restaurant in China’s capital is now scanning customers’ faces and then making menu suggestions based on gender and age. And a popular park in Beijing has deployed smart intelligence to fight toilet paper theft in public restrooms by using face-scanning dispensers that limit each person to a limited amount of paper every nine minutes, the newspaper claimed.
-
- surveillance
- surveillance network
-
(and 2 more)
Tagged with:
-
U.S. regulators ditch net neutrality rules as legal battles loom and Posted From: https://www.yahoo.com/finance/news/federal-communications-commission-set-reverse-111440509.html By David Shepardson WASHINGTON (Reuters) - The U.S. Federal Communications Commission voted along party lines on Thursday to repeal landmark 2015 rules aimed at ensuring a free and open internet, setting up a court fight over a move that could recast the digital landscape. The approval of FCC Chairman Ajit Pai's proposal marked a victory for internet service providers like AT&T Inc, Comcast Corp and Verizon Communications Inc and hands them power over what content consumers can access. Democrats, Hollywood and companies like Google parent Alphabet Inc and Facebook Inc had urged Pai, a Republican appointed by U.S. President Donald Trump, to keep the Obama-era rules barring service providers from blocking, slowing access to or charging more for certain content. Consumer advocates and trade groups representing content providers have planned a legal challenge aimed at preserving those rules. The meeting was evacuated before the vote for about 10 minutes due to an unspecified security threat, and resumed after law enforcement with sniffer dogs checked the room. New York Attorney General Eric Schneiderman, a Democrat, said in a statement he will lead a multi-state lawsuit to challenge the reversal. He called the vote "a blow to New York consumers, and to everyone who cares about a free and open internet." FCC Commissioner Mignon Clyburn, a Democrat, said in the run-up to the vote that Republicans were “handing the keys to the Internet” to a “handful of multi-billion dollar corporations.” Shares of Alphabet, Apple Inc and Microsoft Corp moved lower after the vote. Pai has argued that the 2015 rules were heavy handed and stifled competition and innovation among service providers. "The internet wasn’t broken in 2015. We weren’t living in a digital dystopia. To the contrary, the internet is perhaps the one thing in American society we can all agree has been a stunning success," he said on Thursday. The FCC voted 3-2 to repeal the rules. NEXT STEPS Consumers are unlikely to see immediate changes resulting from the rule change, but smaller startups worry the lack of restrictions could drive up costs or lead to their content being blocked. Internet service providers say they will not block or throttle legal content but that they may engage in paid prioritization. They say consumers will see no change and argue that the largely unregulated internet functioned well in the two decades before the 2015 order. Democrats have pointed to polls showing a repeal is deeply unpopular and say they will prevail in protecting the rules, either in the courts or in U.S. Congress. FCC Commissioner Jessica Rosenworcel, a Democrat, said in a written dissent released on Thursday that the decision grants internet providers "extraordinary new power" from the FCC. "They have the technical ability and business incentive to discriminate and manipulate your internet traffic. And now this agency gives them the legal green light to go ahead," she said. Several state attorneys general said before the vote they would work to oppose the ruling, citing problems with comments made to the FCC during the public comment period. Other critics have said they will consider challenging what they consider to be weaker enforcement. Net neutrality supporters had rallied in front of the FCC building in Washington before the vote. The 2015 rules were intended to give consumers equal access to web content and prevent broadband providers from favoring their own content. Pai proposes allowing those practices as long as they are disclosed. Michael Powell, a former FCC chairman who heads a trade group representing major cable companies and broadcasters, told reporters earlier this week that internet providers would not block content because it would not make economic sense. "They make a lot of money on an open internet," Powell said, adding it is "much more profitable" than a closed system. "This is not a pledge of good-heartedness, it's a pledge in the shareholders' interest." The chief executive of USTelecom, a lobbying group that represents internet providers and the broadband industry, said in a statement the industry has "renewed confidence to make the investments required to strengthen the nation's networks and close the digital divide, especially in rural communities." A University of Maryland poll released this week found that more than 80 percent of respondents opposed a repeal. The survey of 1,077 registered voters was conducted online by the Program for Public Consultation at the University of Maryland from Dec. 6-8. (Reporting by David Shepardson; Writing by Chris Sanders; Editing by Jonathan Oatis and Meredith Mazzilli)
-
- net neutrality
- internet neutrality
-
(and 1 more)
Tagged with: